The operation and maintenance of equipment shall be monitored and managed according to the requirements of verification and documents. Ensure that the operating parameters of the pharmaceutical water purification system are within the range of operating parameters confirmed by the equipment.
The water purification system in pharmaceutical industry is based on the preparation equipment, storage, and export of purified water system and water for injection system and other functional units, optimized and combined by various functional modules, so as to ensure the high performance and quality of the whole system and make the product water fully meet or exceed the water quality standards of purified water and water for injection plant.
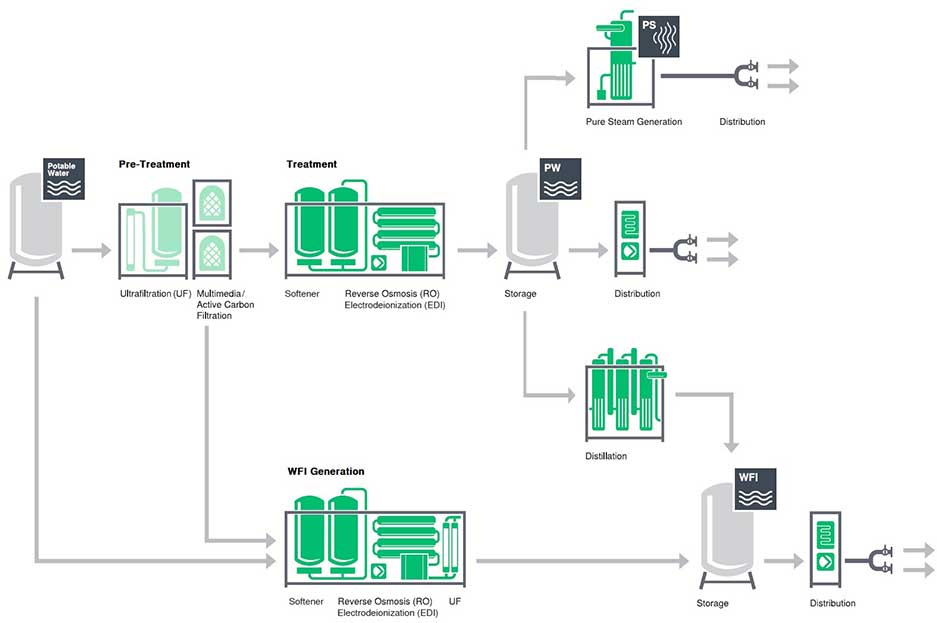
In the pharmaceutical industry, a water purification system is a system designed to remove impurities and contaminants from water to ensure that it is of high quality and meets stringent regulatory standards. Water is a crucial component in the manufacturing process of pharmaceutical products, and its purity is critical to the safety and efficacy of the final product.
The water purification systems in pharmaceutical industry employ various techniques to remove impurities and contaminants from the water. These techniques may include reverse osmosis, deionization, ultrafiltration, electro-deionization, distillation, ozonation, and ultraviolet radiation.
The specific type of water purification system used depends on the quality of the raw water source and the intended use of the water in the manufacturing process. For example, water used for injection (WFI) must meet even higher purity standards than water used for other manufacturing processes.
Water purification systems in the pharmaceutical industry are subject to strict regulations, and the water must be regularly monitored and tested to ensure that it meets the required quality standards. By employing these pharmaceutical water purification systems, pharmaceutical companies can ensure that the water used in their manufacturing processes is of the highest quality and meets all regulatory requirements, thereby ensuring the safety and efficacy of their products.

Fully automatic welding mode is adopted, meeting the special requirements of the Pharmacopoeia.
Intelligent automatic control helps to save labor costs. Modular design mode saves more floor space for you.
24-hour online monitoring to ensure that every drop of treated water is qualified.
Reverse Osmosis (RO) Systems: This technology uses a semi-permeable membrane to remove dissolved solids and impurities from water. RO systems are often used to purify raw water sources, such as well water or surface water, before further treatment.
Deionization (DI) Systems: This technology removes ions from water by passing it through an ion exchange resin. DI systems are typically used to remove dissolved minerals and other impurities from water.
Ultrafiltration (UF) Systems: UF technology uses a semi-permeable membrane to remove suspended solids, bacteria, and viruses from water. It is commonly used in the pharmaceutical industry to remove endotoxins from water.
Electrodeionization (EDI) Systems: EDI technology combines deionization and electrochemistry to remove impurities from water. It is commonly used to produce high-purity water for manufacturing processes.
Distillation Systems: Distillation involves boiling water and condensing the resulting steam to produce pure water. It is commonly used to produce high-purity water for injection systems.
Ozone Systems: Ozone is a powerful oxidizing agent that can destroy bacteria, viruses, and other microorganisms in water. It is commonly used to disinfect water.
UV Systems: Ultraviolet (UV) radiation can destroy microorganisms in water. UV systems are commonly used to disinfect water in the pharmaceutical industry.
Phone:
E-mail:
Address:
Room 1904, Building 10, No. 218, Jiqingmen Street, Jianye District, Nanjing, Jiangsu, China


