The distribution system is the core unit of the whole pharmaceutical water system, and its main function is to ensure that the pressure, flow and temperature delivered to the process pipeline meet the process production requirements. The system uses online monitoring instruments such as flow, pressure, temperature, conductivity and TOC to conduct safety performance, real-time monitoring and trend analysis of water quality, and effectively control the load of microorganisms in water through periodic disinfection or sterilization. In addition, sampling valves shall be installed on the water supply starting point, water pipeline and end pipeline of the entire distribution system for water quality sampling. The whole system is mainly composed of inverter, exchanger, sampling valve, diaphragm valve, sanitary pipe fittings, online monitoring instrument and other supporting control systems.
◆Integrated design, simple operation, and convenient management.
◆Modular structure, saving space.
◆ Ensure that there is turbulence in the circulation pipeline loop,
and the flow rate is>1m/s.
◆Frequency conversion control, the circulation pipeline circuit maintains positive pressure.
◆Online monitoring of temperature, pressure, flow and other technical indicators, alarm and discharge of unqualified water.
◆Operation, disinfection and online monitoring functions are integrated
◆ Equipment manufacturing meets GMP verification requirements.
◆Provide 4Q electronic verification documents.
| DOCUMENT | DOWNLOAD |
Water for Injection Skid | DOWNLOAD PDF |
WFI skid stands for "Water for Injection skid." It is a pre-engineered and pre-assembled system used in pharmaceutical manufacturing to produce Water for Injection (WFI), which is a type of highly purified water used in the production of injectable drugs.
The WFI skid typically consists of a series of components that work together to produce and distribute WFI in a safe and controlled manner. These components may include a multi-stage water purification system, a distillation unit, a storage tank, a distribution pump, and various sensors and controls to monitor and maintain water quality.
The WFI skid is designed to meet the strict regulatory requirements for pharmaceutical manufacturing, including compliance with the United States Pharmacopeia (USP) and European Pharmacopoeia (EP) standards. Its use can help pharmaceutical companies ensure the safety and purity of their injectable drug products, and streamline their production processes.
The correct design of the storage and distribution system is crucial for the success of the injection water system. Our injection purified water storage and distribution system can accurately achieve the following three objectives: to ensure that the quality of injection water is within the scope of the pharmacopeia requirements; Annotate
Injecting water is transported to each process usage point at a flow rate, pressure, and temperature that meets production requirements: ensuring a reasonable allocation of initial investment and operating costs.
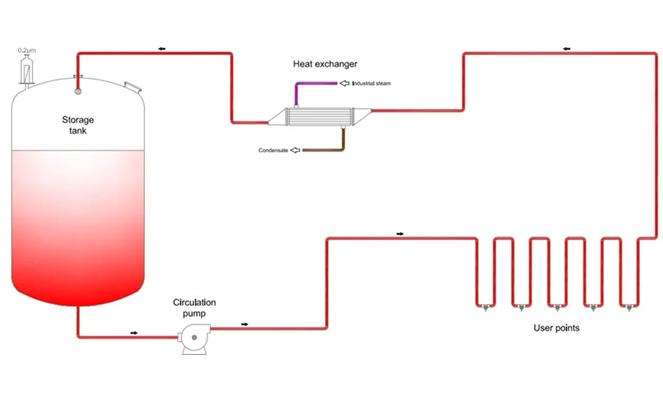
Water for injection storage and distribution systems should be periodically sterilized at the temperature of ≥ 121 ° C. The WFI storage and distribution systems may be sterilized using superheated water or pure steam.
Phone:
E-mail:
Address:
Room 1904, Building 10, No. 218, Jiqingmen Street, Jianye District, Nanjing, Jiangsu, China